|
9/3/2023 0 Comments Medical equipment e1399gylt In other words, the critical part of the asset notion is that its cost cannot be deducted immediately from the company’s revenue, but must be depreciated during its expected useful life. Most dictionaries, including Merriam Webster, define equipment as something like, “all the fixed assets other than land and buildings of a business enterprise.” Moreover, finance professionals classify “fixed assets” as “a company’s tangible, noncurrent assets that are used in its business operations.” And the word “fixed” indicates that these assets will not be consumed or sold in the current accounting year,” according to AccountingCoach. TJC has adopted the FDA definition of “medical devices” and offers the following definition for medical equipment: “ Fixed and portable equipment used for the diagnosis, treatment, monitoring, and direct care of individuals.” Unfortunately, this definition is not helpful either, because it requires defining equipment before determining if the equipment is also medical. The third critical AHJ is The Joint Commission (TJC)-the largest accreditation organization deemed by CMS. Since CMS enforces the Condition of Participation (CoP), which states that “facilities, supplies, and equipment must be maintained to ensure an acceptable level of safety and quality” (42 CFR 482.41(c)(2)), one would expect a clear definition from CMS. Unfortunately, the CMS S&C 14-07 memorandum (CMS, 2013) only offers some examples in the following statement: “… medical equipment, which are devices intended to be used for diagnostic, therapeutic, or monitoring care provided to a patient by the hospital (e.g., IV infusion equipment, ventilators, laboratory equipment, surgical devices, etc.).” So, the key to defining medical equipment lies in deciding what to do with borderline devices.ĬMS is another AHJ since it is the gatekeeper of Medicare and Medicaid reimbursements. 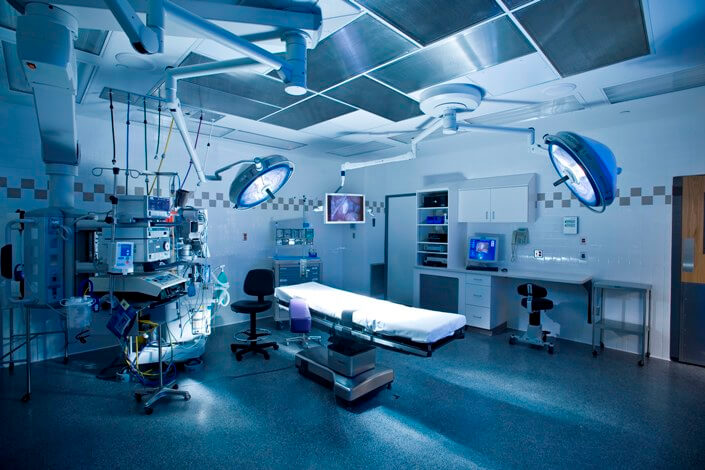
However, there are many “borderline” devices-such as surgical instruments, flowmeters, hand-held pulse oximeters, stethoscopes, sphygmomanometers, rigid endoscopes, etc.-that may last only one to three years and do not need routine, scheduled maintenance instead, they only require occasional repairs or replacement.įurthermore, many of these borderline devices are not individually serialized, making their inventory and service documentation impractical. For example, implants and single-use devices are clearly not equipment that needs to be inventoried or repaired occasionally, as they leave the institution with the patient or are discarded after use. Intuitively, all medical equipment are medical devices-but not all devices should be considered equipment. However, since there is no need for the FDA to distinguish equipment from devices, it is likely that the agency will continue to define everything as a “device” if it “does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of its primary intended purposes.” FDA has a long and complex definition of medical devices because the Food, Drug & Cosmetic Act mandates it to cover devices ranging from tongue depressors to proton beam therapy systems. The reason confusion persists is because no authority having jurisdiction (AHJ) has provided a definition for medical equipment thus far. 
Recently, it has resurfaced even more often, apparently due to some overzealous surveyors trying to strictly enforce the following rule contained in the Centers for Medicare & Medicaid Services’ (CMS’) S&C 14-07 memorandum (CMS, 2013): “All hospital facility and medical equipment, regardless of whether it is leased or owned, and regardless of whether it is maintained according to manufacturer recommendations or is in an AEM program, i s expected to be listed in an inventory, which includes a record of maintenance activities.” Unfortunately, they’ve never come to a consensus. What is medical equipment? At first glance, you would think that this would be an easy question to answer, but the query of what makes a piece of equipment “medical” is one that is debated among clinical engineering/HTM professionals every few years. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
